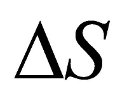
Chapter III, Section A. The entropy in economic and
environmental reasoning
Our economic and environmental sustainability models are commonly
flawed by a lack of understanding, awareness, incorporation, or
even acknowledgment of the reality of entropy. This is not
surprising because the concept of entropy embodies the second law
of thermodynamics, of all the laws of physics, perhaps the hardest
to understand. As a phenomenon, entropy can be defined as the
tendency for all matter and energy in the universe to move to a
state of disorder, the disorder specifically defined as a state of
inert uniformity. The inertness is due to a lack of energy
available to do work. In a closed system not exchanging energy
with its surroundings (or at least another system) the lack of
available energy itself is due to the uniformity of temperature.
Temperature itself is the measure of the internal energy within
the system, but that energy cannot be tapped without a contrast in
temperature. In the thermodynamic sense, entropy is a measure of
the disorder or randomness in a system, AND a corresponding
measure of the amount of unavailable energy. A change in a
system results in a change in entropy, a quantification of the
natural phenomenon of ordering versus disordering, AND energy
becoming available versus unavailable. In the strictest
sense, a change in entropy happens.
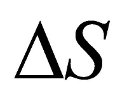
The entropy phenomenon itself is where the conceptualization and
understanding lie, and where its omission leads to fallacious
reasoning. Rifkin best puts the concept of entropy into words:
“Entropy is the measure of the extent to which available energy in
any subsystem of the universe is transformed into an unavailable
form.” (Rifkin; p. 46) Part of the problem incorporating entropy
into our reasoning is in understanding that the second law of
thermodynamics is really two statements, the first readily
understood and at least somewhat incorporated into our economic
and environmental models, and the second somewhat vague, even
among seasoned scientists, and too commonly neglected.
The first statement of the second law of thermodynamics is an
energy statement, that not all energy from a resource can be
converted into useful work, and thus although “energy is neither
created nor destroyed” as “it just changes form”–the basic
statement of the first law–some useful energy is lost in the
conversion, quite literally dissipated. As Rifkin puts it, “Energy
can only be transformed in one direction, from usable to unusable,
from available to unavailable.” Thus a resource of energy must
always be found. To be clear, the entropy of an open system, one
that can receive energy from its surroundings, can be reduced and
energy made available, but it takes an energy input from another
system to do this, and the net for the universe is to move toward
unavailable energy. This concept is the easier 2nd law statement
to understand, lending itself right away to concepts of
efficiency, i.e. the work produced versus the total energy spent,
and people have at least an intuitive sense that we constantly
need energy resources.
The other statement of the second law is a disorder statement, not
so easy to understand, and for many, even hard to accept
philosophically: there is a corresponding universal tendency of
systems to move toward disorder unless a source of energy is
available to maintain or create order. As Rifkin puts it, “Energy
can only be transformed in one direction, from ordered to
disordered.” To be clear, the entropy of an open system, one that
can receive energy from its surroundings, can be reduced and moved
toward order, but it takes an energy input from another system to
do this, and the net for the universe is to move toward disorder.
This concept is the harder of the 2nd law statements to
understand, that energy is required to maintain order, comes from
another system, and that other system MUST become more disordered
than the order generated in the receiving system. People have an
intuitive sense that things move to disorder, a familiarity with
Murphy’s law that what can go wrong will go wrong, but generally
not the sense that somewhere, something MUST "go wrong."
It is important to note that both the energy and disorder
statements of the second law are described in terms of a system,
its surroundings, and the universe which is the sum of the two.
The result of changing entropy within systems is that the entropy
of the universe, the net, is always increasing and never
decreases. The two statements are related, together comprising the
complete concept of entropy, and can be proven mathematically to
be identical. Thus entropy is both a measure of the amount of
energy made unavailable, and a measure of the disorder in a
system, related concepts in that disorder is readily quantifiable
in terms of energy.
Neither economists nor environmentalists consistently understand,
or even fully accept this duality in the phenomenon of entropy.
What is usually neglected in their reasoning is the disorder,
created somewhere else as a consequence of their “solution” to a
problem. The physical reality that entropy happens, that in terms
of its measure, the net change in entropy is always toward
unavailable energy and disorder, is overlooked because the
“solution” applies to the system receiving order, neglecting the
consequences to the surroundings. The system receiving order is
receiving energy, and its boundaries are the limits of what is
being studied or “solved.” The common fatal flaws in both economic
and environmental reasoning are hidden in the erroneous
assumption, realized or not, that this influx of energy that
maintains the system is perpetual, and that the influx comes with
no consequences.
The disregard of entropy can be recognized in economic and
environmental fallacies. Often the disregard is intentional, a
refusal to admit entropy’s reality, because to do so would expose
a fatal flaw in the conceptual system being politically promoted.
In economics, the fatal flaw is the starting assumption that
growth is unlimited, or at least that current levels of wealth are
immutable, even in the face of entropy’s constant tug toward
disorder and decay. In environmentalism, the fatal flaw is a
denial that life itself obtains its order from energy, energy
derived from its surrounding ecosystem, leaving the surroundings
in some way more disordered.
The second law is perhaps a scientific statement of a form of
“original sin.” Life itself MUST create disorder in its
surroundings in order to exist. Thus, ironically, as critical as
the surrounding ecosystem is to our own life support, ultimately a
choice is made between promoting human life versus disrupting the
surrounding ecosystem. This is the root of the fallacies we’ll
discuss next, not only the neglect of the unavailable energy and
disorder left elsewhere, but even if realized, an assumption that
it means there’s no choice. As we’ll later see, the absolution of
this “original sin” is the fact that the surrounding ecosystem is
receiving energy from its surrounding solar system, and thus can
be moved toward order.
 |
 |
 |